Medical Design and Outsourcing
for High Accuracy Mechatronics

Concept to
Product design
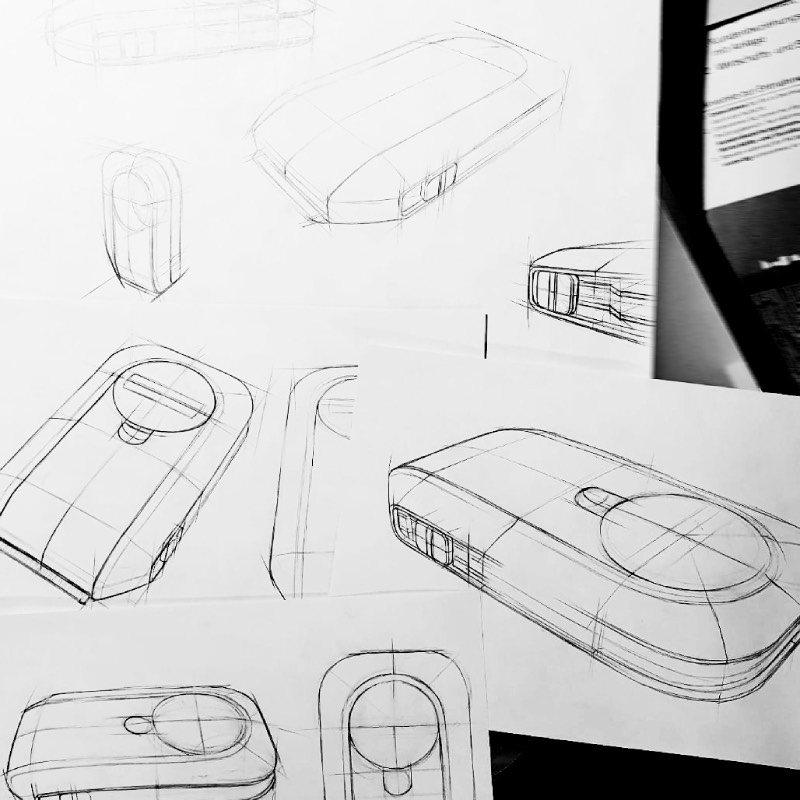
From early ideation visuals
to refined & fully realizable industrial design

Feasibility &
Risk Insight
Material, mechanics, & regulatory considerations
built in early

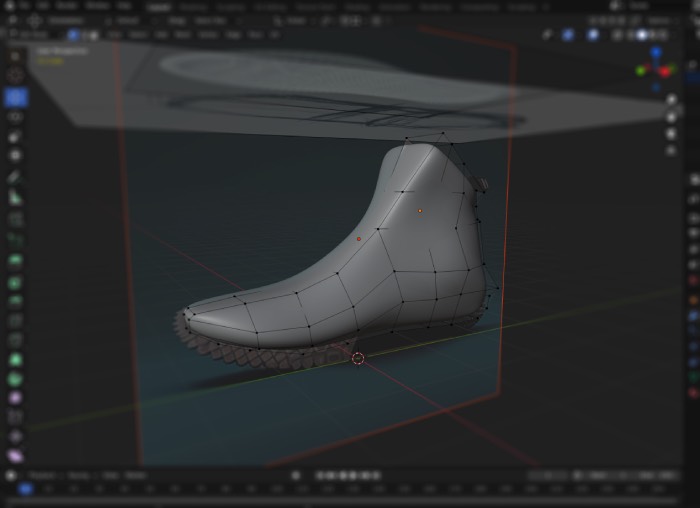
Rapid Prototyping
3D printing & CNC validation
for faster decisions

Human-Centered Design
Creative, intuitive, & ergonomically optimized product usability
What is Medical Design and Outsourcing?
Medical design and outsourcing involves a strategic partnership where an OEM utilizes external expertise for the research and development and manufacturing of healthcare technology. This discipline transforms a clinical idea into a validated product that meets international medical standards such as ISO 13485. By delegating complex mechatronic tasks, companies combine technical functionality with strict safety requirements while focusing on their core clinical competencies.MedTech design is more than aesthetics – it determines usability, compliance, and clinical reliability. Fragmented workflows between design, engineering, and production often cause rework, higher costs, and regulatory delays. Design as a Service eliminates these gaps through unified industrial design and engineering integration.
The Process of Medical Design and Outsourcing
1
Visual Grounding
2
Technical Modeling
3
Physical Prototyping
4
Certification Readiness
Medical design solutions of gKteso
- RPS Positioning Platforms: These systems provide submillimeter patient alignment for stereotactic radiotherapy.
- Beam Shaping Units: Tailored multileaf collimators manage radiation field geometry for specific clinical protocols.
- Ophthalmology Laser Beds: Ergonomic motion units support LASIK and microsurgery through smooth and vibration free control.
- Diagnostic QA-Tools: Tissue equivalent phantoms enable accurate dose calibration and system commissioning.
Advantages of Medical Design and Outsourcing
Accelerated Launch
Outsourcing reduces technical risks and shortens the path to global market entry.
Regulatory Accuracy
Integrated risk management ensures all documentation meets ISO 13485 standards.
Synchronized Workflows
A unified design to engineering process eliminates expensive rework during later stages.
Scalable Output
In house assembly and design transfer facilitate a seamless move to series production.
Operational Clarity:
Realistic visuals and rapid prototypes provide a grounded vision for technical leads.
Material Longevity
Selecting radiation hard or MR safe materials from the start ensures the durability of imaging hardware.
Why gKteso Elevates Your Product Lifecycle
Proven Compliance
Deep Mechatronics
Modular Customization
Unified Ownership
Navigating the Global MedTech Market with Outsourced Engineering





Get in Touch
Bring Your MedTech Product Vision to Life
Contact us to learn how Design as a Service transforms MedTech concepts into ergonomic, regulatory-ready products: from first sketch to full-scale production.
- From first sketch to CE/FDA-ready, production-ready devices
- Seamless integration with your existing R&D and supply chain
- Over 30 years of experience in MedTech hardware & engineering
Request Your Technical Design Quote for the Design of Medical Devices
Plan your next medical device project with an engineering partner who understands mechatronic complexity. Request a consultation with our project engineers or ask for technical data sheets for our modular platforms. We provide a detailed offer for your specific OEM requirements including the full medical design and outsourcing service from concept to series.Design as a Service integrates directly with OEM R&D and engineering teams. Advanced CAD modeling, digital simulation, and interface planning ensure that every design is manufacturable, cleanroom-ready, and regulatory-compliant.
Prototypes undergo usability and ergonomic validation before transfer to scalable production. Materials are selected for radiation compatibility, biocompatibility, and long-term durability. The result: MedTech products that unite functional performance, regulatory reliability, and visual identity in one cohesive design language.
Learn more about our design and engineering solutions for OEM partners
Discover how gKteso’s Design as a Service supports global MedTech companies in creating compliant, innovative, and manufacturable devices. From early ideation to validated production, we help OEMs realize new products faster and more reliably.

Learn more about …
Turning complexity into Clarity – with gKteso’s Design as a Service
Schedule an appointment now!

FAQ: Frequently asked questions for medical design and outsourcing
How does medical design and outsourcing shorten approval times?
Integrated documentation and risk management files ensure your product remains audit ready from the initial design phase. This proactive approach reduces the likelihood of regulatory delays during CE, FDA submissions.
What are the key standards for medical design and outsourcing?
Who manages the design transfer in medical design and outsourcing?
Can you customize existing modules within medical design and outsourcing?
Explore Our Contract Manufacturing Capabilities

Contract Manufacturing
Developed for medical technology innovators and OEMs, our Contract Manufacturing Services turn complex device concepts into certified, market-ready products. From rapid prototyping to validated large-scale production, we ensure regulatory compliance, precision, and supply chain reliability

Customized MLC
Tailored for OEMs, research centers, and novel radiotherapy platforms, our customized MLCs are co-developed to meet unique geometric, electronic, or integration needs. We support you from CAD concept to validated hardware, whether for FLASH, MRI-based therapy, compact systems, or proton beamlines.

Customized OEM QA tools
Customized OEM QA Phantoms and Water Phantom Transport Units from gKteso provide tailored solutions for commissioning, routine QA, imaging calibration, and OEM system integration. With reproducible precision, MDR/FDA compliance, MR compatible options and scalable manufacturing, they ensure reliability in clinical and research environments worldwide.